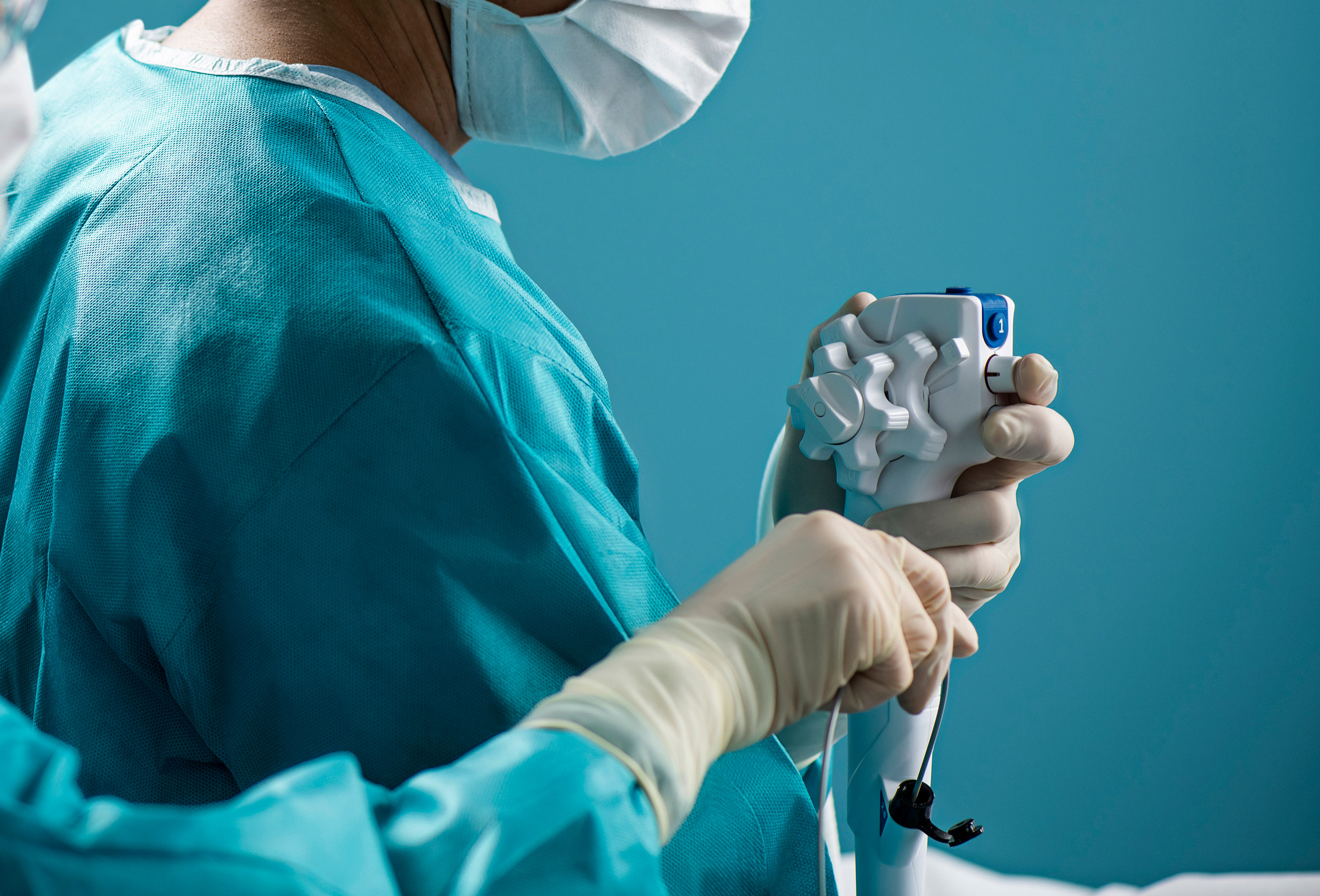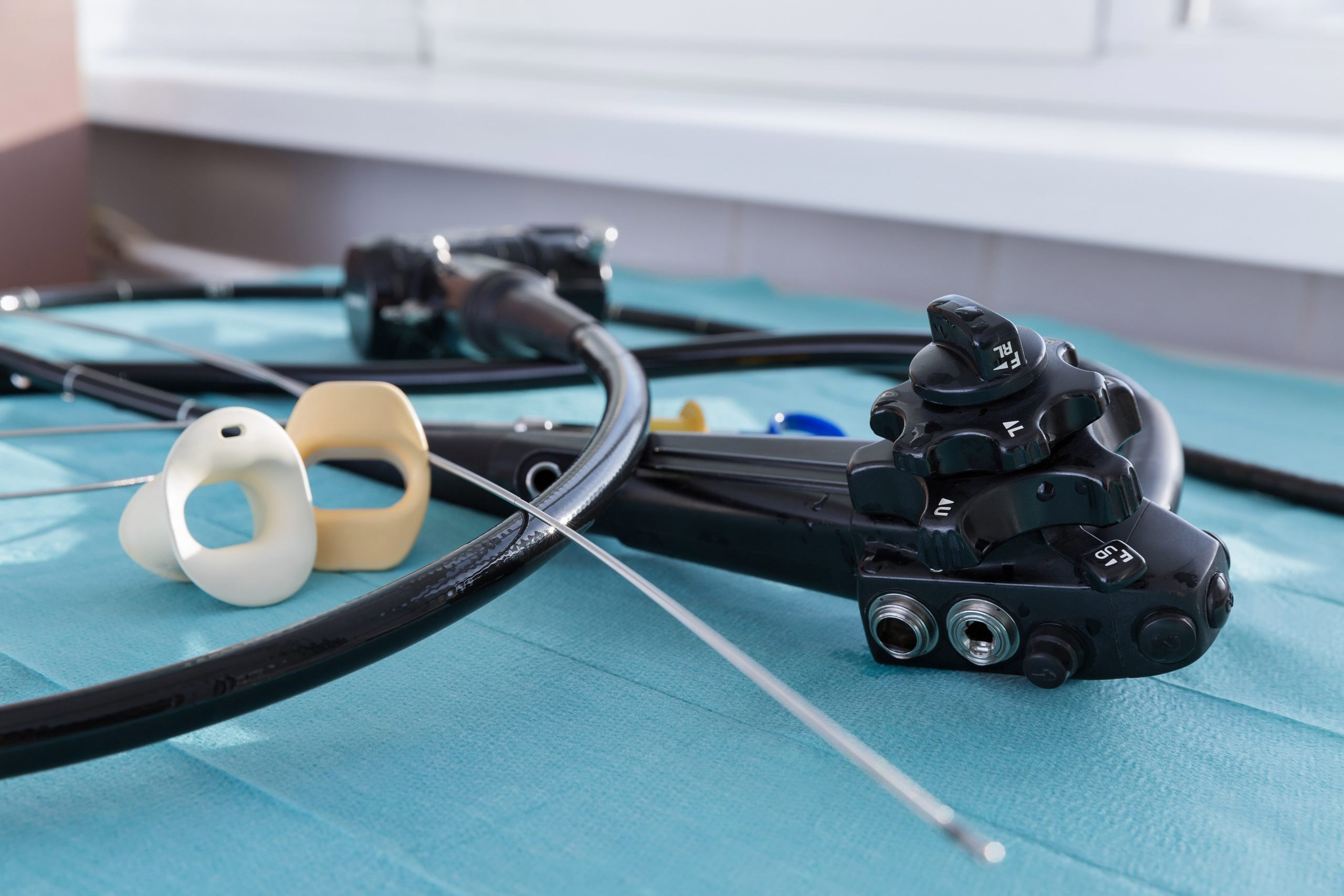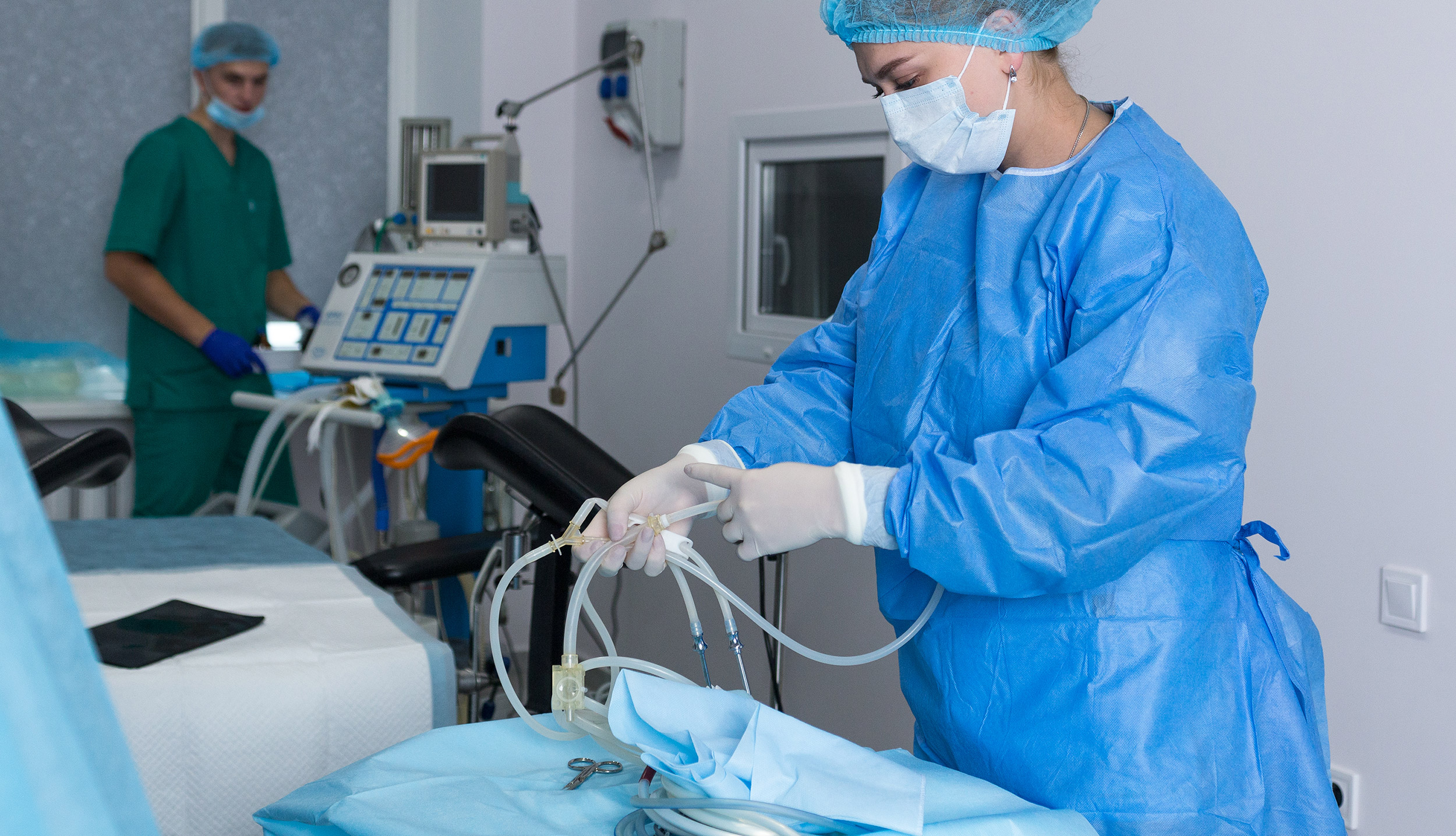
As hospital endoscopy procedure volumes rise with no corresponding staff increase, the added pressure on personnel makes errors in tracking and tracing reusable endoscopes more likely.
So says Suneil Mandava, founder and CEO of Mobile Aspects, who shared his ideas on a recent Beyond Clean podcast entitled “Target Acquired: What is Really Required for Endoscope Tracking & Tracing.” Mandava, a biomedical engineer, started a Pittsburgh-based company that helps top academic and community hospitals track endoscopes through radio frequency identification (RFID) technology, a touchless system that uses radio waves to identify objects.
“The number of procedures is going up dramatically and every hospital is being asked, really forced, to do more procedures with the same number of people, so the stress on these people is very high,” Mandava says. “There’s a doctor screaming, ‘Get the scope over to me,’ so I’m going to make sure I get that done first, then if I have time, I’ll go back and do the documentation. … It’s not a recipe for success.”
Mandava brings personal experience as a Stage 4 cancer survivor who underwent scoping when he was hospitalized for six weeks a few years ago. His company’s system uses sensors and RFID to help track and reduce the number of touches to instruments.
“The cleaning process in between using those endoscopes is very onerous , very complicated, very difficult,” he says. “The designers of these scopes did not make it easy on the hospitals and the clinical teams. … It is a multistep, very tedious, laborious process.”
For hospitals that use reusable scopes, the challenge of cleaning produces a secondary issue of documenting that cleaning. It’s a process that is tracked largely by hand using pen and paper methods, he says.
That kind of system is highly subject to error, especially when people are in a hurry. And sometimes tracking which scope was used for which patient doesn’t occur until the device reaches the cleaning room, once the procedure has been completed, Mandava says.
“When that Joint Commission surveyor comes in and points to a scope and says, ‘Show me all patients that you’ve used it on in the last 90 days,’ a scramble starts to happen,” he adds. “The idea is we really have to move that into 2020-2021 technology and make it electronic and make it really easy to have that information organized.”
Reusable scopes are required to be cleaned on a regular basis even if the scopes haven’t been used, because contamination can still occur. Many hospitals have a seven-day rule, while others allow 14 and, in some cases, 30 days before the endoscopes must be run through a cleaning cycle.
Although some scope manufacturers are working to make it easier to clean hard-to-access parts of scopes, the process will take years to evolve, Mandava says. In the case of duodenoscopes, the FDA already has said “the best solution to reducing the risk of disease transmission by duodenoscopes is through innovative device designs that make reprocessing easier, more effective, or unnecessary.”
Single-use devices eliminate all the tracking, tracing and reprocessing required with reusables. Boston Scientific’s disposable duodenoscope became the first to be cleared for marketing by the U.S. Food and Drug Administration in December 2019. Ambu A/S received 510(k) clearance from the FDA for its single-use duodenoscope in July 2020. Ambu launched the world’s first single-use flexible bronchoscope in 2009.
Click here to listen to the entire Beyond Clean podcast and you can find its episode library here.