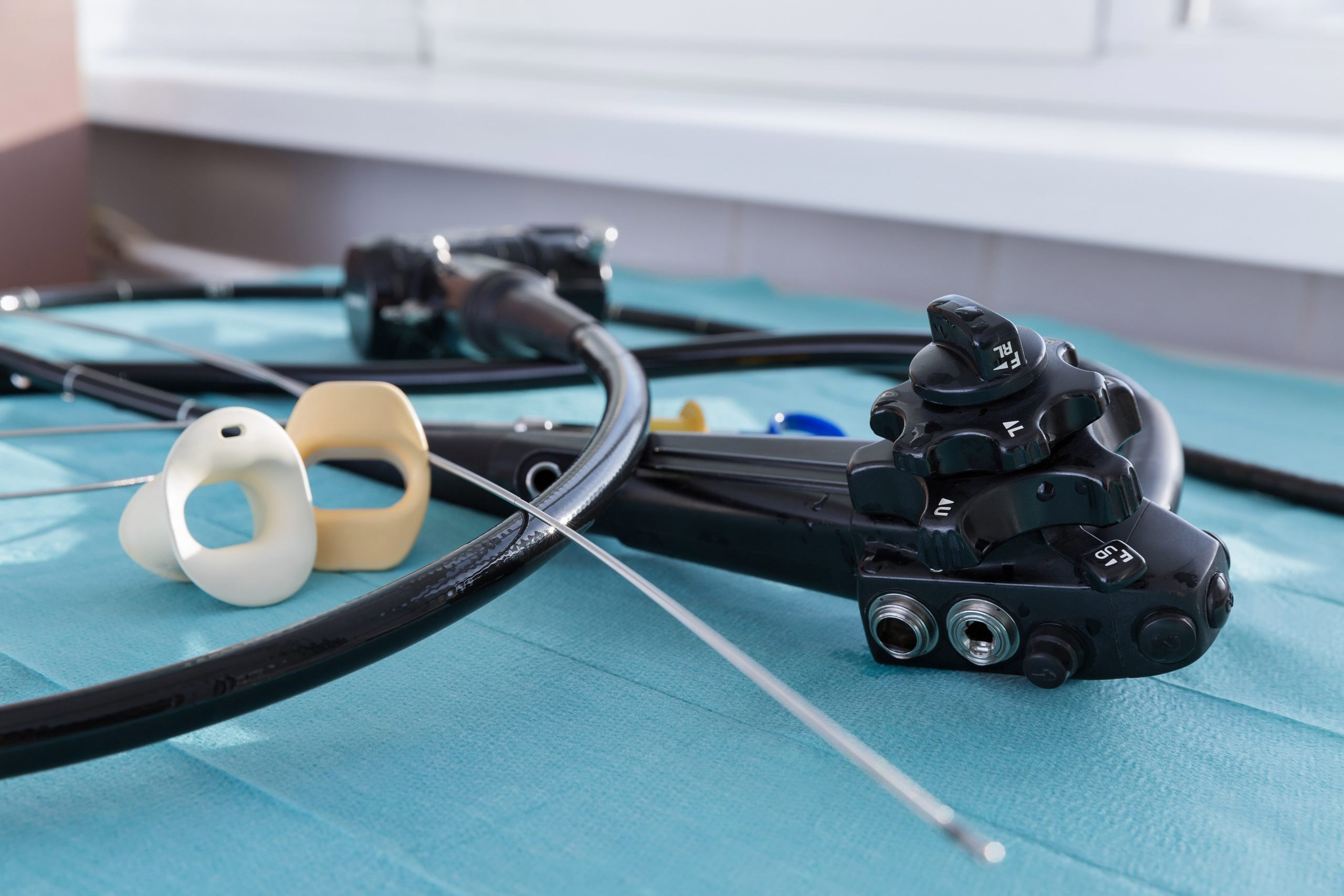
The U.S. Food and Drug Administration has updated a safety communication on duodenoscopes and is recommending that hospitals and endoscopy facilities “complete transition to innovative duodenoscope designs” due to concerns about reprocessing and patient cross-contamination.
The FDA previously said hospitals should consider using duodenoscopes with disposable components or adopt fully single-use models. The agency issued this new update in part because of new postmarket surveillance studies on fixed endcap design duodenoscopes that found 4.1 percent to 6.6 percent of samples tested positive with what the agency calls high-concern organisms.
Emerging data “suggests that the best solution to reducing the risk of disease transmission by duodenoscopes is through innovative device designs that make reprocessing easier, more effective, or unnecessary,” the communication says. “Disposable designs may reduce between-patient duodenoscope contamination by half or more as compared to reusable, or fixed endcaps.”
Duodenoscopes are used in more than 500,000 endoscopic retrograde cholangiopancreatography (ERCP) procedures annually in the U.S. Their complex design makes them particularly hard to clean. Failure to properly reprocess endoscopes can, in rare instances, result in patient-to-patient disease transmission caused by fluid or tissue remaining after cleaning.
The FDA has cleared five duodenoscopes that feature disposable components and two fully single-use duodenoscopes, including one from Ambu A/S that won approval in July 2020 and was also granted Breakthrough Device Designation. Fixed endcap duodenoscopes are no longer marketed in the U.S. and those that are still in use at healthcare facilities should be replaced, according to the safety communication. Some manufacturers are offering replacement programs to upgrade fixed endcap scopes.
The alert urges patients or caregivers to not cancel or delay any planned procedures without first discussing it with their provider. Hospitals and endoscopy facilities should ensure that staffers are “meticulously following reprocessing instructions,” the FDA said.
This means instituting a quality control program that includes sampling and microbiological culturing, considering reprocessing with sterilization or a liquid chemical sterilant processing system, and monitoring reprocessing procedures. Instructions to do so properly can be found here.
The FDA update includes new information on completed postmarket surveillance studies, also known as 522 studies. The high-concern organisms identified in the studies are defined as organisms that are more often associated with disease, such as E. coli, and Pseudomonas contamination after reprocessing.
The ECRI Institute recently published its annual list of the top 10 health technology hazards, which included poor duodenoscope reprocessing ergonomics and workflows that put healthcare workers and patients at risk. Correcting these problems will require, among other strategies, “reevaluating the use of reusable duodenoscopes,” according to the report. Failure to adequately reprocess contaminated duodenoscopes between uses is known to spread deadly pathogens, according to new ECRI survey data. The risks of injury to reprocessing staffers and how ergonomic and workflow factors can compromise reprocessing effectiveness and put patients at risk are not as well known.
The update came the same day the agency issued a letter to healthcare providers detailing a voluntary recall or urological endoscopes by medical device manufacturer Karl Storz. The company is instructing customers to discontinue high-level disinfection and, in some cases, liquid chemical sterilization of its urological endoscopes. Testing of the manual high-level disinfection process showed that required efficacy levels were not reached.
Update: The FDA updated the communication on June 30, 2022 to include a recent culturing and sample study completed by Fujufilm which found its newer model duodenoscope with a disposable componenet to have no samples test positive for enough low concern organisms to indicate a reprocessing failure and only 1.1 percent test positive with high concern organisms. The FDA’s conclusion remained the same, that use of the newer models of duodenoscopes can reduce the risk of infection for patients, compared to the older fixed endcap duodenoscope models.