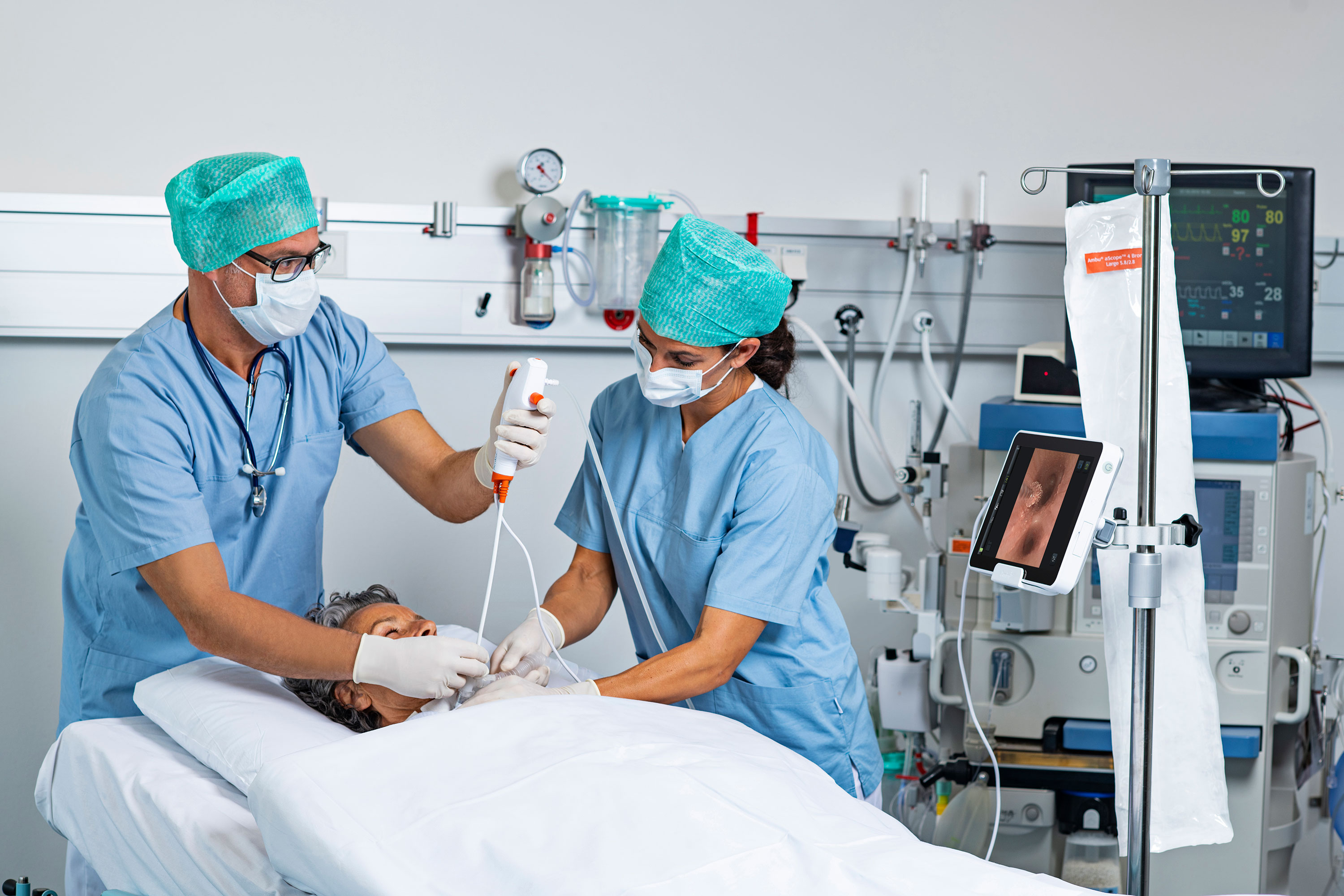
Eliminating risk of patient cross-contamination is a key reason why some U.S. gastroenterologists are willing to use a single-use duodenoscope for up to half of endoscopic retrograde cholangiopancreatography (ERCP) procedures.
This is according to a recent anonymous survey of 70 U.S. gastroenterologists performing ERCP — one of the most challenging endoscopic procedures. The results were first published in a poster at the American College for Gastroenterology conference in October 2020.
“It gave a nice indication that there is a willingness to try single-use,” said Sara Larsen, a global health economist at Ambu A/S and lead author of the abstract. “I think this is very promising.”
A previous meta-analysis Larsen conducted found duodenoscopes may have contamination rates of up to 15.25 percent — worth noting during Patient Safety Awareness Week, which runs from March 14 to 20, 2021. This is an Institute for Healthcare Improvement initiative to raise awareness about preventable harm in the healthcare space.
Duodenoscopes contain three long, narrow channels and an elevator mechanism at the distal end, offering plenty of real estate for microorganisms to thrive. In some recorded cases, reprocessing these flexible endoscopes has proven ineffective at preventing all potential for patient infection. Single-use duodenoscopes, however, are made to use once and discard and thus eliminate risk of cross-contamination.
Larsen and her team conducted the survey of U.S. gastroenterologists just three months after the U.S. Food and Drug Administration granted 510(k) clearance for the world’s first single-use duodenoscope. That means gastroenterologists were commenting on a switch to a product that most of the market did not have access to.
Given this reality, the team was surprised at the results, Larsen told Single-Use Endoscopy.
The U.S. physicians surveyed said the top reasons for considering a switch to single-use for 50 percent of ERCP cases were cost transparency and minimizing risk of infection. In fact, the U.S. gastroenterologists reported an interest in converting 54 percent of ERCPs to single-use if contamination rates at their facilities were reported to be greater than 10 percent for patient-ready scopes.
The surveyors collected answers anonymously with an online survey tool, QuestionPro. The reason why gastroenterologists may convert only half of procedures were not discussed in the published research.
The research team posed the same survey questions to 105 gastroenterologists performing ERCPs in France, Germany and the United Kingdom. European physicians indicated they would, on average, convert 39 percent of ERCP procedures to single-use.
U.S. physicians at facilities where reusable duodenoscopes were owned, rather than leased, indicated a greater willingness to convert to single-use duodenoscopes. The opposite proved true in the European survey.
Larsen speculated the difference in potential conversion rates for U.S. and European physicians likely had to do with Boston Scientific Corp. — the first company to create a single-use duodenoscope — being an American company. Ambu A/S, based in Ballerup, Denmark, has released a single-use model since the publication of this research. The FDA granted 510(k) clearance to the aScope Duodeno in July 2020.