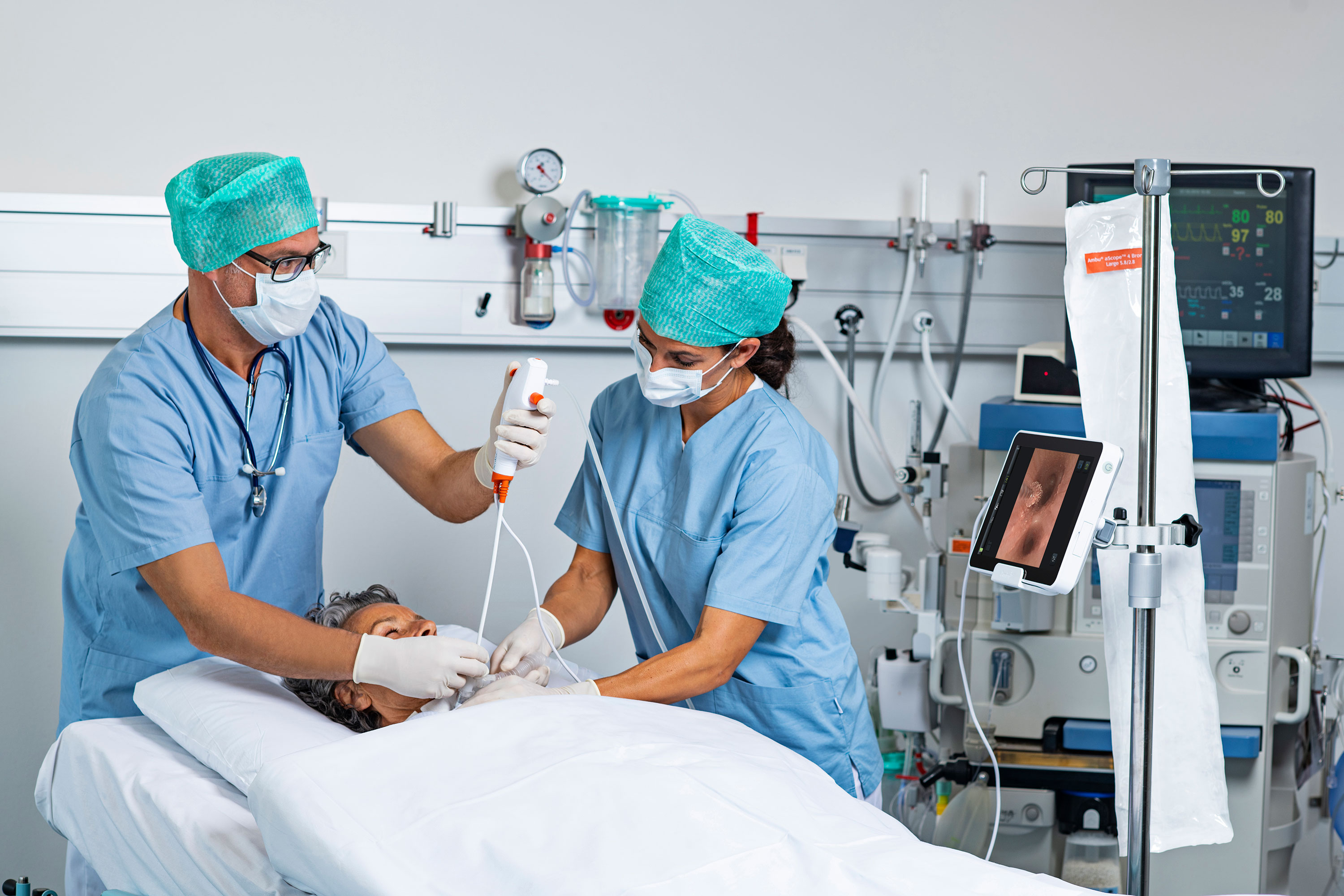
The safety net that single-use bronchoscopes bring to intensive care was never more evident than during the early days of the global pandemic in New York City.
"Patients were clogging tubes with secretions left and right," Dr. James M. Horowitz, a cardiologist and critical care specialist, said in a recent webinar hosted at the Becker’s Hospital Review 11th Annual Meeting. “A team of surgeons scoped room after room. They did hundreds of bronchoscopies and none of them contracted COVID-19."
"There's no way we could have kept up without deploying disposable scopes,” he added. His intensive care unit at NYU Langone Health used Ambu single-use bronchoscopes throughout the pandemic.
Horowitz was joined in the Becker's webinar by two other physicians who discussed the benefits of single-use endoscopy in their areas of specialty. The three shared firsthand accounts of the patient safety, financial, and workflow benefits of single-use endoscope technology.
Another key selling point, they said, is the quick turnaround time for technological advancement.
Typically, reusable endoscopes and image processing systems have very long product life cycles. And that leads to slower adoption of new technologies — because expensive equipment sits on the balance sheet, only getting replaced when it fails.
"My bronch tower is like a VCR,” Horowitz said. “It doesn't have Wi-Fi and it doesn't connect to my EMR. …This is becoming an increasingly important regulatory compliance issue with diagnosis and billing."
In addition, the COVID-19 pandemic has vastly heightened infection awareness. With a return to elective procedures, patients are even starting to request single-use endoscopes, the physicians said in the webinar.
The U.S. Food and Drug Administration last month updated a safety communication on reprocessing flexible bronchoscopes and is recommending that healthcare providers consider using single-use bronchoscopes when there is increased risk of spreading infection or when immediate reprocessing is not available.
Previous FDA post-market surveillance studies of flexible duodenoscopes point to several reasons for inconsistencies in reprocessing success and subsequent cross-contamination risks. The complexity of reprocessing such delicate instruments, as well as time pressures on technicians, may cause vital steps to be skipped or performed inadequately, the studies found.
The FDA is encouraging healthcare providers to consult a March 2020 American Association for Bronchology and Pulmonology (AABIP) COVID-19 safety communication for guidance on when to use single-use bronchoscopes.
In addition to the safety and convenience that single-use endoscopes offer, clinicians recognize potential financial advantages.
“When [hospital administrators] compared six figures in repair costs for reusable equipment with the cost of single-use scopes, the decision was a no-brainer,” Dr. Srinivas Vourganti, a urologist at Rush Medical Center, said during the Becker’s session.